|
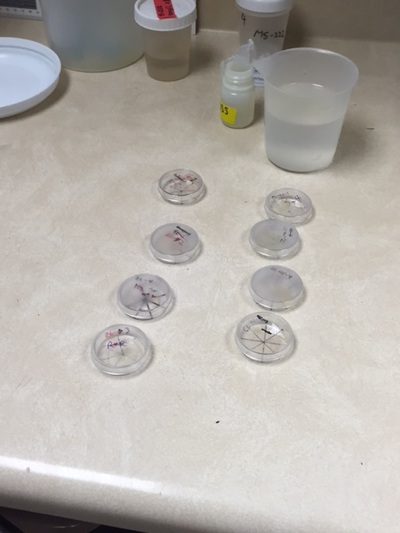
With school starting, I wasn't able to go to OSU often throughout the week. I only went two days: Monday and Saturday. With my rigorous schedule in the upcoming school year along with my after school activities like tennis practice and going to the gym, I will most likely only be able do research on the weekends. In my two days at the Lab, I continued monitoring the zebrafish mating behaviors and fertility counts. I used my time outside of the lab to input the data onto the computer. Next week, I plan to summarize all the data we have gathered so far to identify any trends. On Saturday, we noticed something interesting. Normally, the triploid zebrafish do not spawn the same amount of eggs as normal, diploid zebrafish. However, on 8/20 the heat-shocked zebrafish were found to produce more eggs than usual, with almost no eggs left in the fish's ovary. The images below show the large amount of eggs. My schedule will begin to clutter as school progresses. Hopefully, I will be able to continue my research on zebrafish fertility throughout my senior year!
0 Comments
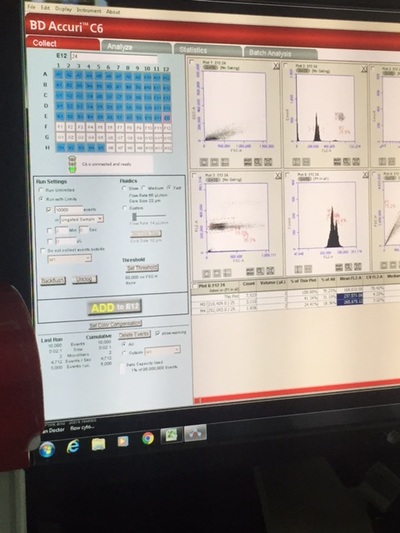
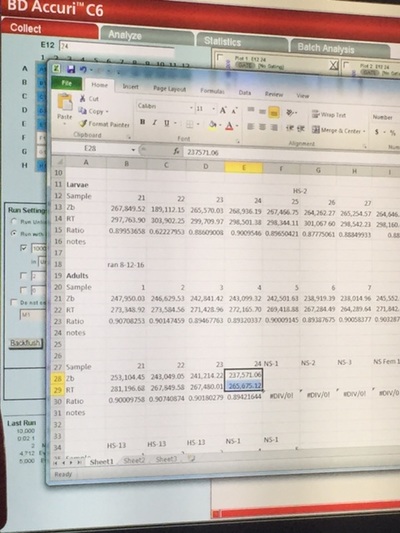
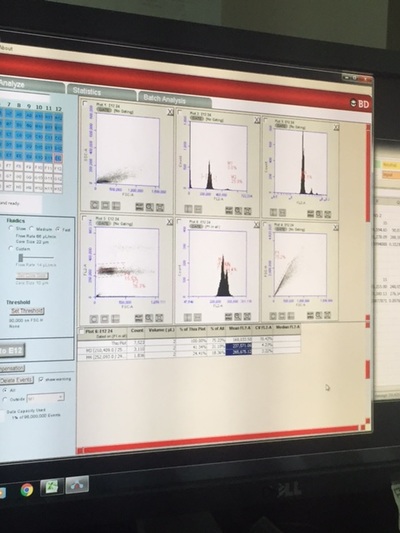
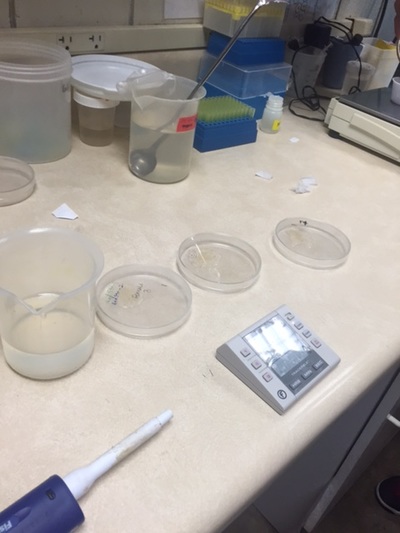
In my second week of internship at OSU, I continued to monitor the spawning behavior of triploid zebrafish and continued to record the fertilization and 24 hour survival of the eggs. Similar to last week, I had a very structured schedule Monday through Wednesday. From 7:30 to 10:30, I observed zebrafish spawning behavior. It is important that we allow the fish to mate at this time because of their unique circadian rhythm. After the fish stopped demonstrating mating behavior, we pulled the female fish out of the tank and stripped them of their remaining eggs to perform "in vitro fertilization", which is artificial fertilization that occurs outside of the organism. I would then check for the fertilization count in both the natural (in vivo) spawn and artificial (in vitro) spawn. The video demonstrates one of the key spawning behaviors that occurs throughout the entire mating process: male chase female. The gender of adult zebrafish can be identified by their body shape. Female's are generally more round than males because of the eggs stored in their ovary. We needed to give the fish time to rest after their 3 days of mating, so we decided to take a break from observing the spawning behaviors. This gave me a good opportunity to upload all of my information into the computer. This proved to take a long time because of the multiple different formats I had to use to input the information. It took the majority of the day. Afterwards, I helped clean the some fish tanks. On Friday, I helped with another project: Studying the effect of different nutrition on zebrafish growth. Similar to last Friday, I helped input the growth data into the computer. Afterwards, I helped stain the DNA of the newly spawned zebrafish cells. My student mentor and I went to the Veterinary Medicine Lab to use their Flow Cytometer, which measures the fluorescence in each cell. Since the DNA was stained with a fluorescent stain, the amount of fluorescence is proportional with the amount of DNA. This allows us to evaluate the type of ploidy in each cell. What we found was very interesting. Each of our 24 samples were triploid. I go back to school on Wednesday, so I will not be able to observe the zebrafish spawning behaviors because I will be at school during the 7:30-10:30 time period. Hopefully I will be able to continue to work at the lab in the after school on some days.
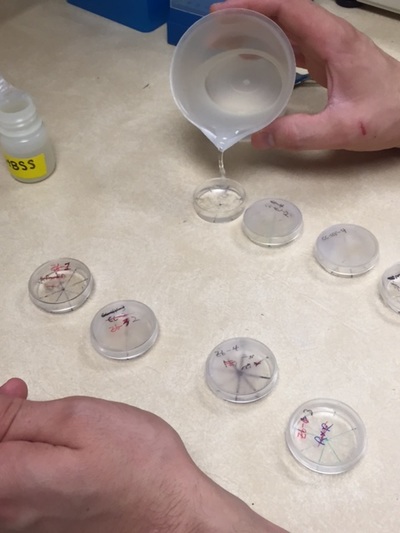
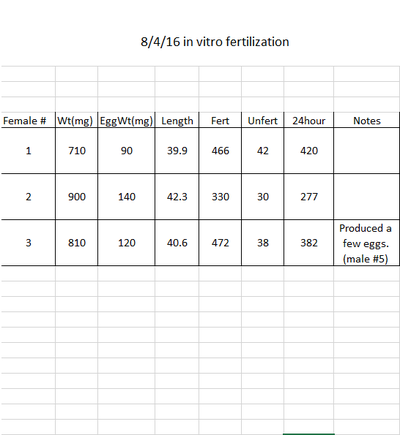
Going into my first week of internship at OSU, I didn't know what to expect. I wasn't sure if I was going to wash petri dishes or cleaning aquariums. However, I was pleasantly surprised that Dr. Dabrowski, professor of environmental science, had planned for me to do research. To assist me, he found articles that would benefit me and my research. I found that these articles were extremely useful, and they helped me understand the background of my upcoming research: studying the effect of triploidy on zebra fish fertility. My first three days of research were very similar. For the first two hours, I would observe 3 groups fish mate and write down each observation as well as the time it took place. After the fish stopped showing mating behaviors, I would spend the next hour cleaning fish tanks around the lab. This hour was necessary to allow the eggs produced by the fish to fertilize. After the eggs were given sufficient time, I would examine the eggs of each aquarium and calculate the number of fertilized and unfertilized eggs, as well as record the length and weight of the male. It would take up to two hours to get an accurate record of the eggs and fish characteristics. Lastly, I would once again use the microscope to observe the eggs from the previous day and record the amount of eggs that survived the first 24 hours. The other days did not have a consistent theme. Sometimes, I would begin my day by preparing food for the zebra fish and zoo-plankton. On Thursday I started my first "in vitro fertilization", which means that instead of having two fish mate and produce eggs (in vivo fertilization), the sperm and egg were collected and fertilized outside of the body. I also recorded the the male weight and length, as well as the fertilization count and the 24 hour survival. I also helped with other projects in the lab. On Friday, I helped a nutrition experiment, in which the fish were given different types and combinations of food to see which tank would show the most optimal growth. My mentor Thomas Delomas and I recorded the weight and length of all the fish in the tank. The results of the experiment would be most accurate if each fish tank had the same amount of fish, so we also counted all the fish and transferred the "extra" fish to spare tanks in the greenhouse. My first week was very stressful, but my I gradually became less nervous as the week progressed. Once I got over my nerves, the research became very enjoyable. I wonder what Week 2 has in store for me.Time to read more articles on zebra fish!
|
Archives
February 2017
Categories |












 RSS Feed
RSS Feed
